Human papillomavirus (HPV) and cervical cancer
Human papillomavirus (HPV) is a group of viruses that are extremely common worldwide.

What is HPV?
Human papillomavirus (HPV) is the most common viral infection of the reproductive tract. Most sexually active women and men will be infected at some point in their lives and some may be repeatedly infected.
The peak time for acquiring infection for both women and men is shortly after becoming sexually active. HPV is sexually transmitted, but penetrative sex is not required for transmission. Skin-to-skin genital contact is a well-recognized mode of transmission.
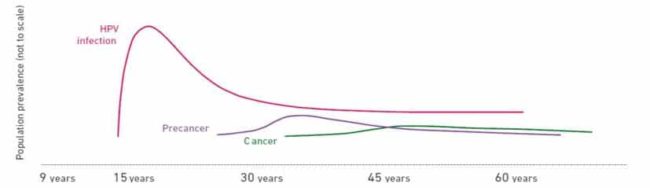
There are many types of HPV, and many do not cause problems. HPV infections usually clear up without any intervention within a few months after acquisition, and about 90% clear within 2 years. A small proportion of infections with certain types of HPV can persist and progress to cervical cancer.
Cervical cancer is by far the most common HPV-related disease. Nearly all cases of cervical cancer can be attributable to HPV infection.
The infection with certain HPV types also causes a proportion of cancers of the anus, vulva, vagina, penis and oropharynx, which are preventable using similar primary prevention strategies as those for cervical cancer.
Non-cancer causing types of HPV (especially types 6 and 11) can cause genital warts and respiratory papillomatosis (a disease in which tumours grow in the air passages leading from the nose and mouth into the lungs). Although these conditions very rarely result in death, they may cause significant occurrence of disease. Genital warts are very common, highly infectious and affect sexual life.
How HPV infection leads to cervical cancer
Although most HPV infections clear up on their own and most pre-cancerous lesions resolve spontaneously, there is a risk for all women that HPV infection may become chronic and pre-cancerous lesions progress to invasive cervical cancer.
It takes 15 to 20 years for cervical cancer to develop in women with normal immune systems. It can take only 5 to 10 years in women with weakened immune systems, such as those with untreated HIV infection.
Risk factors for HPV persistence and development of cervical cancer
- HPV type – its oncogenicity or cancer-causing strength;
- immune status – people who are immunocompromised, such as those living with HIV, are more likely to have persistent HPV infections and a more rapid progression to pre-cancer and cancer;
- coinfection with other sexually transmitted agents, such as those that cause herpes simplex, chlamydia and gonorrhoea;
- parity (number of babies born) and young age at first birth;
- tobacco smoking
Global burden of cervical cancer
Worldwide, cervical cancer is the fourth most frequent cancer in women with an estimated 570 000 new cases in 2018 representing 7.5% of all female cancer deaths. Of the estimated more than 311 000 deaths from cervical cancer every year, more than 85% of these occur in low and middle income countries. Women living with HIV are six times more likely to get cervical cancer compared to women without HIV, and an estimated 5% of all cervical cancer cases are attributable to HIV (2).
In high income countries, programmes are in place which enable girls to be vaccinated against HPV and women to get screened regularly. Screening allows pre-cancerous lesions to be identified at stages when they can easily be treated.
In low and middle income countries, there is limited access to these preventative measures and cervical cancer is often not identified until it has further advanced and symptoms develop. In addition, access to treatment of such late-stage disease (for example, cancer surgery, radiotherapy and chemotherapy) may be very limited, resulting in a higher rate of death from cervical cancer in these countries.
The high mortality rate from cervical cancer globally (Age Standardized Rate: 6.9/100,000 in 2018) could be reduced by effective interventions.
Cervical cancer control: A comprehensive approach
The Global strategy towards eliminating cervical cancer as a public health problem adopted by the WHA in 2020, recommends a comprehensive approach to cervical cancer prevention and control. The recommended set of actions includes interventions across the life course.
The life-course approach to cervical cancer interventions
| Primary prevention | Secondary prevention | Tertiary prevention |
|---|---|---|
Girls 9-14 years
|
Women 30 years old or older |
All women as needed |
Girls and boys, as appropriate
|
|
Treatment of invasive cancer at any age
|
It should be multidisciplinary, including components from community education, social mobilization, vaccination, screening, treatment and palliative care.
Primary prevention begins with HPV vaccination of girls aged 9-14 years, before they become sexually active.
Women who are sexually active should be screened for abnormal cervical cells and pre-cancerous lesions, starting from 30 years of age in the general population of women. Screening for sexually active women living with HIV should start at an earlier age, once they have tested positive for HIV.
If treatment of pre-cancer is needed to excise abnormal cells or lesions, cryotherapy or thermal ablation both destroy abnormal tissue on the cervix) is recommended and is performed in outpatient clinic.
If signs of cervical cancer are present, treatment options for invasive cancer include surgery, radiotherapy and chemotherapy and patients need to be referred to the right level of services.
HPV vaccination
There are currently 3 vaccines that have been prequalified, all protecting against both HPV 16 and 18, which are known to cause at least 70% of cervical cancers. The third vaccine protects against five additional oncogenic HPV types, which cause a further 20% of cervical cancers. Given that the vaccines which are only protecting against HPV 16 and 18 also have some cross-protection against these other less common HPV types which cause cervical cancer, WHO considers the three vaccines equally protective against cervical cancer. Two of the vaccines also protect against HPV types 6 and 11, which cause anogenital warts.
Clinical trials and post-marketing surveillance have shown that HPV vaccines are very safe and very effective in preventing infections with HPV infections, high grade precancerous lesions and invasive cancer (3).
HPV vaccines work best if administered prior to exposure to HPV. Therefore, WHO recommends to vaccinate girls, aged between 9 and 14 years, when most have not started sexual activity. The vaccines cannot treat HPV infection or HPV-associated disease, such as cancer.
Some countries have started to vaccinate boys as the vaccination prevents genital cancers in males as well as females, and two available vaccines also prevent genital warts in males and females.
WHO recommends vaccination for girls aged between 9 and 14 years, as this is the most cost- effective public health measure against cervical cancer
HPV vaccination does not replace cervical cancer screening. In countries where HPV vaccine is introduced, screening programmes may still need to be developed or strengthened.
Screening and treatment of pre-cancer lesions
Cervical cancer screening involves testing for pre-cancer and cancer, more and more testing for HPV infection is performed. Testing is done among women who have no symptoms and may feel perfectly healthy. When screening detects an HPV infection or pre-cancerous lesions, these can easily be treated, and cancer can be avoided. Screening can also detect cancer at an early stage and treatment has a high potential for cure.
Because pre-cancerous lesions take many years to develop, screening is recommended for every woman from aged 30 and regularly afterwards (frequency depends on the screening test used). For women living with HIV who are sexually active, screening should be done earlier, as soon as they know their HIV status.
Screening has to be linked to treatment and management of positive screening tests. Screening without proper management in place is not ethical.
There are 3 different types of screening tests that are currently recommended by WHO:
- HPV DNA testing for high-risk HPV types
- Visual inspection with Acetic Acid (VIA)
- conventional (Pap) test and liquid-based cytology (LBC)
For treatment of pre-cancer lesions, WHO recommends the use of cryotherapy or thermal ablation and Loop Electrosurgical Excision Procedure (LEEP) when available. For advanced lesions, women should be referred for further investigations and adequate management.
Management of invasive cervical cancer
When a woman presents symptoms of suspicion for cervical cancer, she must be referred to an appropriate facility for further evaluation, diagnosis and treatment.
Symptoms of early stage cervical cancer may include:
- Irregular blood spotting or light bleeding between periods in women of reproductive age;
- Postmenopausal spotting or bleeding;
- Bleeding after sexual intercourse; and
- Increased vaginal discharge, sometimes foul smelling.
As cervical cancer advances, more severe symptoms may appear including:
- Persistent back, leg and/or pelvic pain;
- Weight loss, fatigue, loss of appetite;
- Foul-smell discharge and vaginal discomfort; and
- Swelling of a leg or both lower extremities.
Other severe symptoms may arise at advanced stages depending on which organs cancer has spread.
Diagnosis of cervical cancer must be made by histopathologic examination. Staging is done based on tumor size and spread of the disease within the pelvis and to distant organs. Treatment depends on the stage of the disease and options include surgery, radiotherapy and chemotherapy. Palliative care is also an essential element of cancer management to relieve unnecessary pain and suffering due the disease.
WHO response
The World Health Assembly adopted the global strategy to accelerate the elimination of cervical cancer as a public health problem and its associated goals and targets for the period 2020–2030 (WHA 73.2) (4). The global strategy to eliminate cervical cancer has set targets to accelerate the elimination:
- a threshold of 4 per 100,000 women-year for elimination as a public health problem
- 90–70–90 targets that need to be met by 2030 for countries to be on the path towards cervical cancer elimination
- 90% of girls fully vaccinated with the HPV vaccine by age 15.
- 70% of women are screened with a high-performance test by 35, and again by 45 years of age.
- 90% of women identified with cervical disease receive treatment (90% of women with pre-cancer treated; 90% of women with invasive cancer managed).
WHO has developed guidance and tools on how to prevent and control cervical cancer through vaccination, screening and management of invasive cancer and a knowledge repository will make them available through a single point. WHO works with countries and partners to develop and implement comprehensive programmes in line w the global strategy.
References
(1) Ferlay J, Ervik M, Lam F, Colombet M, Mery L, Piñeros M, Znaor A, Soerjomataram I, Bray F (2018). Global Cancer Observatory: Cancer Today. Lyon, France: International Agency for Research on Cancer. Available from: https://gco.iarc.fr/today
(2) Stelzle D, Tanaka LF, Lee KK, et al. Estimates of the global burden of cervical cancer associated with HIV. Lancet Glob Health 2020; published online Nov 16. DOI:S2214-109X(20)30459-9 https://www.thelancet.com/journals/langlo/article/PIIS2214-109X(20)30459-9/fulltext
(3) Lei et al. (2020) HPV Vaccination and the Risk of Invasive Cervical Cancer. N Engl J Med 2020;383:1340-8. DOI: 10.1056/NEJMoa1917338
(4) World Health Organization. Global strategy to accelerate the elimination of cervical cancer as a public health problem https://www.who.int/publications/i/item/9789240014107
















